Pharmaceutical Translation Services: Ensuring Regulatory Compliance Across Global Markets
Pharmaceutical translation services convert complex regulatory, clinical, and scientific documentation while maintaining the precision required for global drug approvals. When patient safety and market authorization depend on accurate multilingual documentation, specialized translators with pharmaceutical expertise become essential partners in your regulatory strategy.
Table of Contents
What Pharmaceutical Translation Services Deliver
Pharmaceutical translation encompasses the conversion of highly regulated documentation from source to target languages while preserving scientific accuracy, regulatory compliance, and terminological consistency. Unlike general translation, this specialization requires translators who understand pharmacology, clinical research methodology, and the specific requirements of regulatory authorities worldwide.
The scope extends far beyond simple language conversion. Professional translation services in the pharmaceutical sector must navigate complex terminology databases, adhere to standardized templates mandated by regulatory bodies, and ensure that safety-critical information remains unambiguous across all language versions. A single mistranslated dosage instruction or contraindication can have serious consequences for patient safety and regulatory standing.
Why Pharmaceutical Translation Requires Specialized Expertise
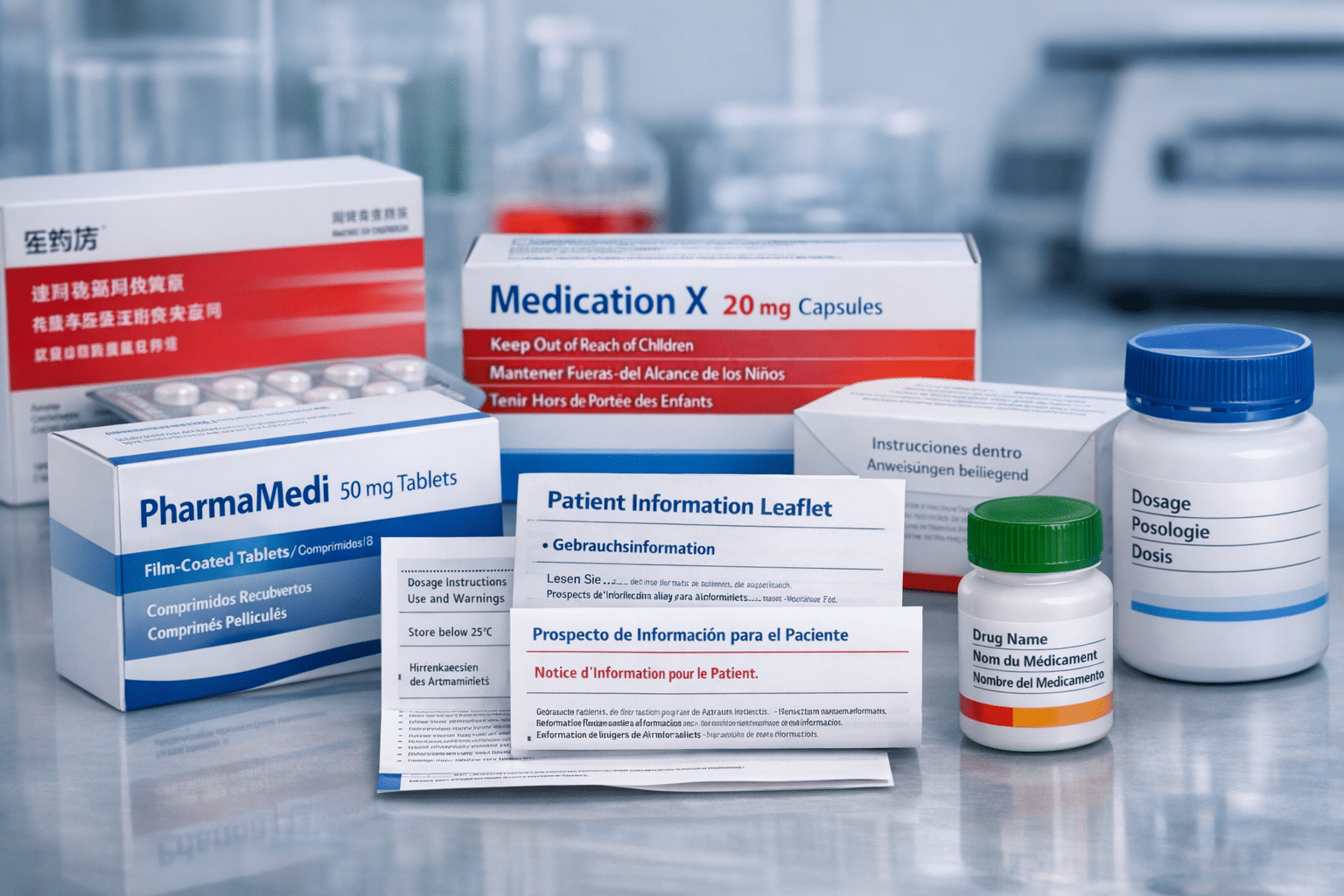
The pharmaceutical industry operates under intense regulatory scrutiny from agencies such as the European Medicines Agency (EMA) and the U.S. Food and Drug Administration (FDA). These bodies require that product information, including the Summary of Product Characteristics (SmPC), Patient Information Leaflets (PILs), and labeling, be translated into the official languages of each market where the drug is authorized.
Translators working in this field must demonstrate competence in medical and pharmaceutical terminology, understand the regulatory frameworks governing drug approval, and maintain strict confidentiality protocols. The ISO 17100 certification provides a framework for verifying translator qualifications and ensuring consistent quality across all projects.
Regulatory Requirements Across Markets
Different regulatory jurisdictions impose specific requirements for pharmaceutical documentation. Understanding these requirements is essential for successful market authorization and ongoing compliance.
The EMA mandates that all product information be available in the official languages of EU member states where the drug is marketed. The agency provides standardized Quality Review of Documents (QRD) templates that must be followed precisely. Following a positive opinion from the Committee for Medicinal Products for Human Use (CHMP), marketing authorization holders have only five days to submit national-language versions of their product information.
The FDA requires that drug labeling in the United States be provided in English, though certain documents may require translation into Spanish for markets with significant Spanish-speaking populations. Clinical trial documentation submitted to the FDA must meet specific formatting and content requirements outlined in the Common Technical Document (CTD) format.
Pharmaceutical companies seeking to enter markets in Japan must submit documentation to the Pharmaceuticals and Medical Devices Agency (PMDA) in Japanese. Similarly, submissions to China's National Medical Products Administration (NMPA) require documentation in Simplified Chinese. Each jurisdiction has unique requirements for terminology, format, and supporting documentation.
Document Types Requiring Specialized Translation
The pharmaceutical translation portfolio encompasses diverse document types, each with specific requirements for accuracy and regulatory compliance.
Regulatory submissions form the foundation of market authorization. These include marketing authorization applications, variation applications, and periodic safety update reports. Each document type follows specific templates and must adhere to precise formatting requirements, and translation for pharmaceutical regulatory submissions is held to tighter terminological and procedural standards than general medical content. Certified translations may be required for official submissions to regulatory authorities.
Clinical trial documentation requires particular attention to accuracy given its direct impact on patient safety and data integrity. Informed consent forms must be translated in language that patients can understand while maintaining legal and ethical requirements. Clinical study protocols, investigator brochures, and case report forms all require precise translation to ensure consistency across multinational trials.
Product labeling and packaging materials represent the final interface between pharmaceutical products and patients. Patient Information Leaflets must communicate complex medical information in accessible language while meeting regulatory requirements for content and format. Carton text, blister packs, and container labels must fit within space constraints while providing complete safety information.
Pharmacovigilance documentation supports ongoing safety monitoring throughout a product's lifecycle. Individual Case Safety Reports (ICSRs), periodic safety update reports, and risk management plans require accurate translation within tight timeframes. The EMA's Good Pharmacovigilance Practices guidelines specify that ICSRs must include either verbatim text from primary sources or accurate translations.
Manufacturing and quality documentation ensure consistent production standards across global facilities. Standard operating procedures, batch records, validation protocols, and quality control reports must be translated accurately to maintain Good Manufacturing Practice (GMP) compliance.
Quality Assurance in Pharmaceutical Translation
Quality assurance in pharmaceutical translation extends beyond linguistic accuracy to encompass regulatory compliance, terminological consistency, and process validation.
The ISO 17100 standard establishes requirements for translation service providers, including translator qualifications, revision processes, and project management procedures. Under this standard, every translation must be revised by a second qualified linguist before delivery. This mandatory second review catches errors that might otherwise compromise patient safety or regulatory compliance.
Terminology management is critical in pharmaceutical translation. Customised glossaries ensure that technical terms are translated consistently across all documentation, from regulatory submissions to patient-facing materials. Translation memory systems store previously approved translations for reuse, maintaining consistency across documents and reducing turnaround times.
Quality control processes in pharmaceutical translation typically include multiple review stages. Initial translation is followed by revision, which may be supplemented by specialized review for regulatory or medical accuracy. Final proofreading ensures that formatting matches regulatory templates and that no errors were introduced during production.
Validation of translation processes may be required for submissions to certain regulatory authorities. This involves documenting translator qualifications, establishing audit trails, and demonstrating that quality management systems meet applicable standards. Companies operating under ISO 13485 for medical devices may extend these requirements to their translation providers.
Selecting a Pharmaceutical Translation Provider
Choosing a pharmaceutical translation provider requires evaluation of multiple factors beyond price and turnaround time. The consequences of translation errors in this sector make provider selection a critical business decision.
Certification to ISO 17100 demonstrates that a translation provider has established quality management systems meeting international standards. This certification requires documented processes for translator qualification, project management, and quality assurance. For pharmaceutical clients, this certification provides assurance that translations will undergo proper review and quality control.
Native translators with subject matter expertise are essential for pharmaceutical translation. Translators should demonstrate competence in pharmacology, clinical research, or regulatory affairs in addition to linguistic qualifications. Experience with specific document types and regulatory frameworks further ensures accurate, compliant translations.
Technology infrastructure supports efficiency and consistency in pharmaceutical translation. Translation memory systems, terminology databases, and quality assurance tools enable providers to maintain consistency across large documentation sets and reduce turnaround times without compromising quality. Integration with regulatory submission systems may also be relevant for large pharmaceutical clients.
Client references and testimonials from pharmaceutical companies provide evidence of provider capability. Experience with specific regulatory authorities, document types, and therapeutic areas indicates relevant expertise. The ability to scale for large submissions while maintaining quality is particularly important for global pharmaceutical operations.
Ready to discuss your pharmaceutical translation requirements? Request a quote to receive a detailed proposal tailored to your regulatory submission timeline and quality requirements.